How to Pick the Best Hydration Drink

The original performance hydration solution was pure water. Although this tasty beverage still works — such as whenever you’re not hammering a workout — it’s not the most effective in situations where you’re sweating or expending energy.
When it comes to hydrating for performance, there’s a lot more to it than picking the grooviest looking tub or grabbing the bottle that your favorite pro drinks.
An enormous amount of science goes into developing the powders and liquids behind the fancy packaging, so selecting the right one can make all the difference between victory and sitting on the sidelines nursing a cramp.
How Hydration Drinks Work
Science has discovered better ways than just water to hydrate during intense exercise, given physical exertion depletes H2O and electrolytes through sweat, blood sugar, and glycogen (a form of sugar stored in your muscles and liver).
It centers on a concept called osmosis. When fluids are separated by a semi-permeable membrane (your gut lining, in this case), the fluid with fewer dissolved particles (meaning it’s less concentrated) will cross the barrier to the fluid with more dissolved particles (meaning it’s more concentrated) in an attempt to restore balance. Take a look at the image below to see what we mean.
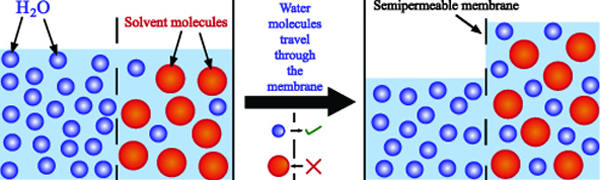
Why does this matter? At first, the budding sports supplement industry came up with a Swiss Army knife solution, creating drinks that not only hydrated, but also replenished those electrolytes and carbs. These drinks fall into three different categories: hypertonic, hypotonic, and isotonic drinks.
Hypertonic drinks
Because many popular hydration beverages contain a lot of sugar (about 15 grams or more per eight ounces of water), they’re considered hypertonic, meaning they have more dissolved particles than your blood. Specifically, they have more than 280 mosmol/kg.
The problem with hypertonic drinks is they have osmosis working against them. The body absorbs the drinks more slowly and they can pull water out of your system and into your intestines. This means they end up dehydrating you in an effort to create a balance between fluids.
What’s more, all that unabsorbed liquid in your stomach can slosh around (yes, “slosh” is actually the term the scientists use), causing gastric upset. If you ever slammed a 16-ounce bottle of neon blue sports drink in the middle of a hard effort and suddenly felt like you were going to lose it (or actually lost it), this is probably why.
Hypotonic drinks
That leads us to hypotonic drinks, in which the sugar concentration is lower than the particle concentration in your blood. The optimal level for these seems to be 200–250 mosmol/kg or around nine grams of sugar per eight ounces of water.
That little bit of sugar helps the body absorb both water and electrolytes — crucial, given that studies indicate that losing even one to two percent of your body mass to dehydration can result in a 10% reduction in performance. The weaker you perform, the worse your results will be.
Also, the small amount of sugar, while intended to aid absorbency, still provides a little fuel, and this isn’t a bad thing.
Isotonic drinks
A third category is isotonic drinks, which have an equal amount of dissolved particles as your blood. Some athletes use these in an attempt to fuel without the issues of hypertonic drinks. If you can’t otherwise consume calories during your activity, this might be a good solution, but according to the research, this form of hydration isn’t as effective as hydrating with hypotonic solutions.
Isn’t the Sugar in These Drinks an Issue?

If you’re concerned about consuming sugar, don’t be. Sugar itself isn’t a problem in our diets. Several healthy foods contain sugar — most notably, almost all fruit. The problems arise from too much sugar. And excess isn’t the case when you consume these types of drinks during hard exercise.
It’s simple math. A solid workout burns 400–800 calories per hour — primarily blood sugar and glycogen. An hour’s worth of a hypotonic sports drink is about 80 calories. You’re good. In fact, if you’re doing a prolonged effort (two hours or more), you’ll want to augment your hydration with more carbs in the form of solid food to prevent bonking.
The performance hydration market is a big one. You have no shortage of options, but the choice you make should be obvious if you consider two factors: How does it taste, and is it effective for your specific needs?
For that second factor, understanding osmolality will narrow the field considerably. Armed with this knowledge, you can move on to the fun part of the selection process: tasting them.
Glossary
Osmosis – When fluids are separated by a semi-permeable membrane (your gut lining, for example), the fluid with less dissolved particles (or “stuff”) will cross the barrier to the fluid with more dissolved particles in an attempt to restore balance.
Osmolality – A measure of the number of dissolved particles in a fluid. Human bodily fluids, including blood, have an osmolality between 280 and 295 milliosmoles per liter (mosmol/kg).
Hypertonic – When a fluid has more dissolved particles. For us, it refers to the sugar concentration in a beverage being higher than the particle concentration in your blood. Hypertonic drinks have an osmolality in excess of 280 mosmol/kg.
Hypotonic – When a fluid has less dissolved particles. For us, it refers to the sugar concentration in a beverage being lower than the particle concentration in your blood.
Isotonic – When a fluid has an equal amount of dissolved particles. In this case, it refers to the sugar concentration in a beverage being the same as the particle concentration in your blood.
